- Inspire
- Innovate
- Integrate
- Home
- Who we are
- Services
Industries
BIM
- BIM Modeling & Digital Delivery
BIM Modeling & Digital Delivery
- BIM Coordination & Constructability
BIM Coordination & Constructability
- BIM Automation & Digital Engineering
BIM Automation & Digital Engineering
- BIM Data, Cost & Handover Services
BIM Data, Cost & Handover Services
- Architectural Design, Visualization & Presentation Support
Architectural Design, Visualization & Presentation Support
- Structural & Steel Detailing Services
Structural & Steel Detailing Services
Engineering Simulation Analysis
Sustainability Services
Digital Services
Acoustic Services
Energy & Intelligence
Outsourcing Technical Experts
Infrastructure
BIM
Outsourcing Technical Experts
Engineering Design
Sustainability
Energy & Utilities
Simulation & Analysis
Engineering Manpower Secondment
Oil & Gas Engineering services
Engineering Simulation Analysis
Engineering Manpower Secondment
Sustainability Services
Energy & Intelligence
Outsourcing Technical Experts
- BIM Modeling & Digital Delivery
- Portfolio
- Training
- Blog
- Careers
- Life at Conserve
- Contact Us
- Home
- Case Studies
- CFD Airflow Cleanliness Validation and Confirms Contaminant Removal in Hospital Operation Theatre
CFD Airflow Cleanliness Validation and Confirms Contaminant Removal in Hospital Operation Theatre
Project Snapshot
Category | Details |
|---|---|
| Client | Hospital operation theatre in Oman |
| Sector | Healthcare |
| Platform / Software | Ansys CFX |
| Standards | ASHRAE 170, ISO 14644-1, HTM 03-01 |
| Division | Conserve Solutions — Simulation & Analysis Division |
| Key Outcome | Cleanliness validated pre-commissioning; unidirectional laminar airflow confirmed above surgical site; contaminant and recirculation risks eliminated at design stage |
Problem Statement
We had designed the ventilation system for our new operation theatres to achieve ISO 5 cleanroom conditions, but we had no way to confirm that the laminar airflow canopy was genuinely protecting the surgical site, or that contaminated exhaust air – including anaesthetic gases – was being effectively captured and removed. We needed simulation to validate containment before the theatre went live.
Impact of Challenges
Patient Safety Risk: Without validated laminar airflow, airborne particulates and micro-organisms could contaminate the surgical field, increasing the risk of surgical site infections (SSIs) — a critical patient safety concern.
Regulatory Non-Compliance: Failure to demonstrate ISO 5 conditions at the surgical site and HTM 03-01 air change rates could prevent theatre licensing and commissioning approval from health authorities.
Undetected Recirculation: Contaminated exhaust air returning toward the surgical field cannot be identified through manual inspection or standard commissioning tests — CFD is the only reliable detection method.
Design Blind Spots: Theatre furniture, equipment trolleys, surgical lights, and the clinical team’s body heat all disturb airflow in ways that standard hand calculations cannot capture — risking ISO classification failure post-installation.
Conserve Solutions - How We Solved It
a. Thinking (Strategy)
Conserve approached this engagement as a clinical risk elimination exercise, not a standard airflow calculation. The primary objective was to provide the infection control and M&E teams with CFD-backed evidence that the OT ventilation design would deliver ISO 5 cleanliness at the surgical site and achieve compliant removal of contaminants including and airborne biological particles – under real operating conditions.
b. Execution (What We Built)
- Developed a high-fidelity 3D CFD model of the operation theatre in Ansys CFX, incorporating the laminar airflow canopy geometry, HEPA supply diffuser array, low-level exhaust grilles, surgical light pendants, operating table, instrument trolleys, anaesthetic machine, and the thermal body heat loads of a person surgical team
- Modelled the laminar airflow canopy as a uniform velocity inlet with HEPA-filtered supply with positive pressure differential relative to the scrub corridor, in line with HTM 03-01 pressure cascade requirements
- Applied turbulence modelling with thermal buoyancy coupling to accurately resolve the complex interaction between cool laminar supply air and the rising thermal plumes from the surgical lights and clinical team’s body heat.
Quantified air change rates at the occupied zone and validated velocity uniformity beneath the LAF canopy against ISO 14644-3 requirements
c. Integration (Impact Layer)
CFD outputs were structured directly against ISO 14644-1 particle class limits and HTM 03-01 directional flow compliance thresholds. The infection control team received annotated contour maps at surgical field level, vector plots showing laminar flow integrity beneath the canopy, and contaminant concentration heat maps identifying any zone where exhaust re-entrainment occurred.
This allowed the ventilation contractor to make targeted adjustments – surgical light pendant positioning and exhaust grille relocation – prior to physical installation, avoiding costly rework.
Project Timeline:
Phase 1 – Day 1-2 | Kick-off, data collection, boundary condition setup, and CFD-compatible 3D model development |
Phase 2 – Day 3-4 | Mesh independence study and CFD simulations. |
Phase 3 – Day 5-6 | Post-processing, ASHRAE validation, and final report submission |
Delivered - What we delivered:
- Laminar airflow velocity uniformity plots – LAF canopy coverage verification against ISO 14644-3 requirements
- Air change effectiveness (ACE) and ventilation effectiveness analysis – quantifying how efficiently fresh air replaces contaminated air within the surgical zone
- Exhaust grille performance validation – capture velocity and extraction efficiency under all scenarios
- Thermal comfort and stratification analysis – evaluation of temperature distribution, draft risk, and compliance with clinical comfort standards
- Design risk register and design optimisation recommendations – surgical light pendant positioning, exhaust grille relocation, and LAF canopy sizing guidance
- Client-ready technical report with executive summary, regulatory compliance matrix, and commissioning readiness statement
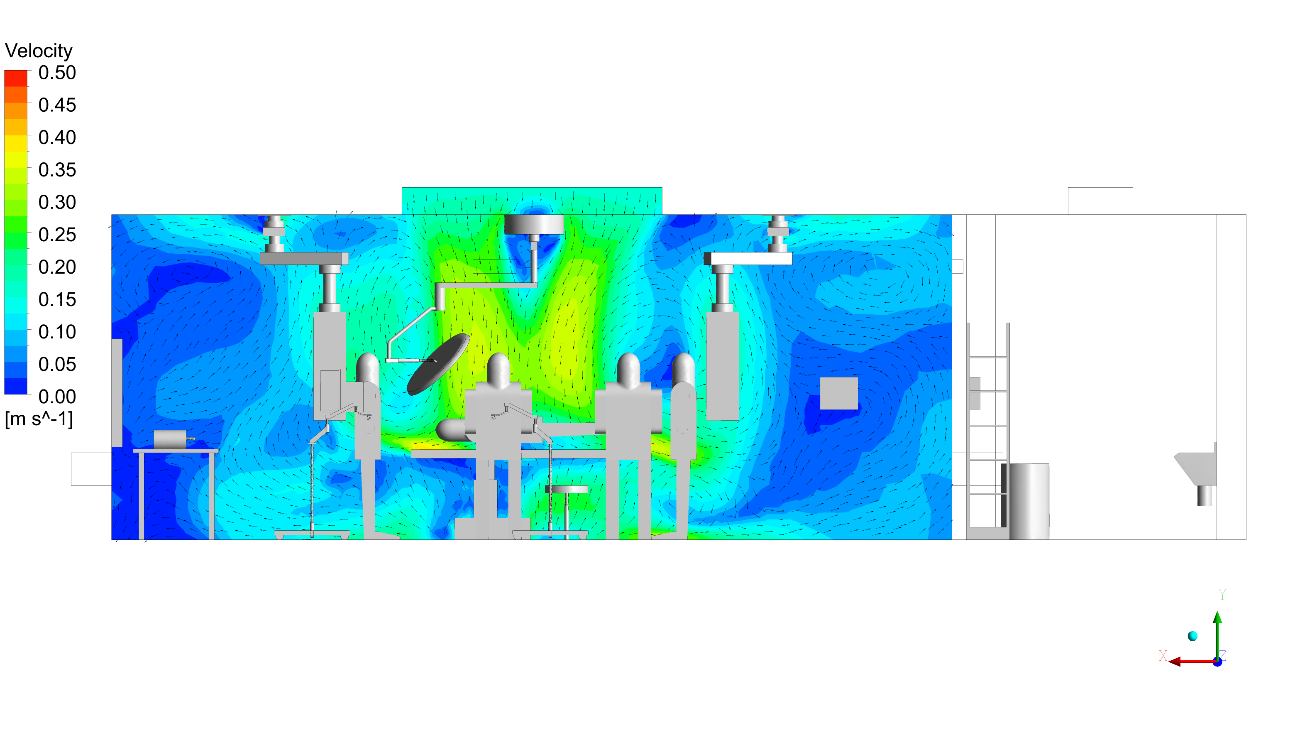
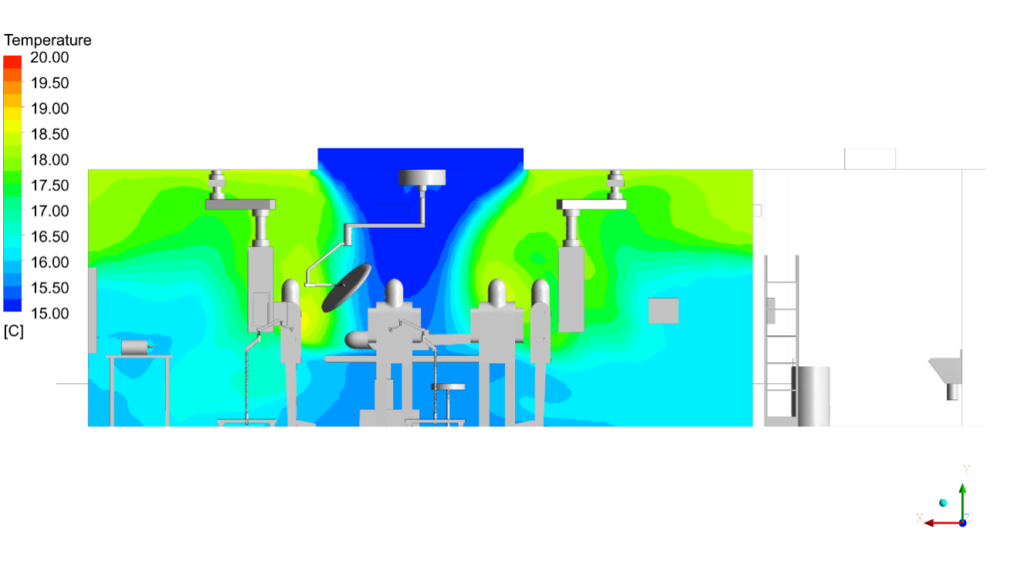
Software and Technology:
Ansys CFX | Primary CFD solver – thermal-fluid coupled simulation for cleanroom airflow, contaminant dispersion, and pressure cascade analysis |
ISO 14644-3 | Cleanroom test methods standard – laminar airflow velocity uniformity and unidirectionality verification criteria |
HTM 03-01 | UK Health Technical Memorandum – ventilation for healthcare premises; directional airflow, pressure cascade, and air change rate requirements for operation theatres |
ASHRAE 170 | Ventilation of Healthcare Facilities – supplementary air quality and ventilation rate standard for surgical environments |
Before vs After
Parameter | Before (Risk) | After CFD Validation | Result |
Surgical Site Air Cleanliness | ISO 5 assumed by design; no verification | ISO 5 cleanliness level confirmed at surgical field under all scenarios | ISO compliant |
LAF Canopy Airflow Uniformity | Uniform airflow assumed; impact of lights and pendants not assessed | Uniform & downward airflow achieved; local disturbances identified and mitigated | Compliant |
Recirculation / Airflow Disturbance | Potential recirculation due to equipment and heat loads | Recirculation zones identified and resolved through equipment repositioning | Risk mitigated |
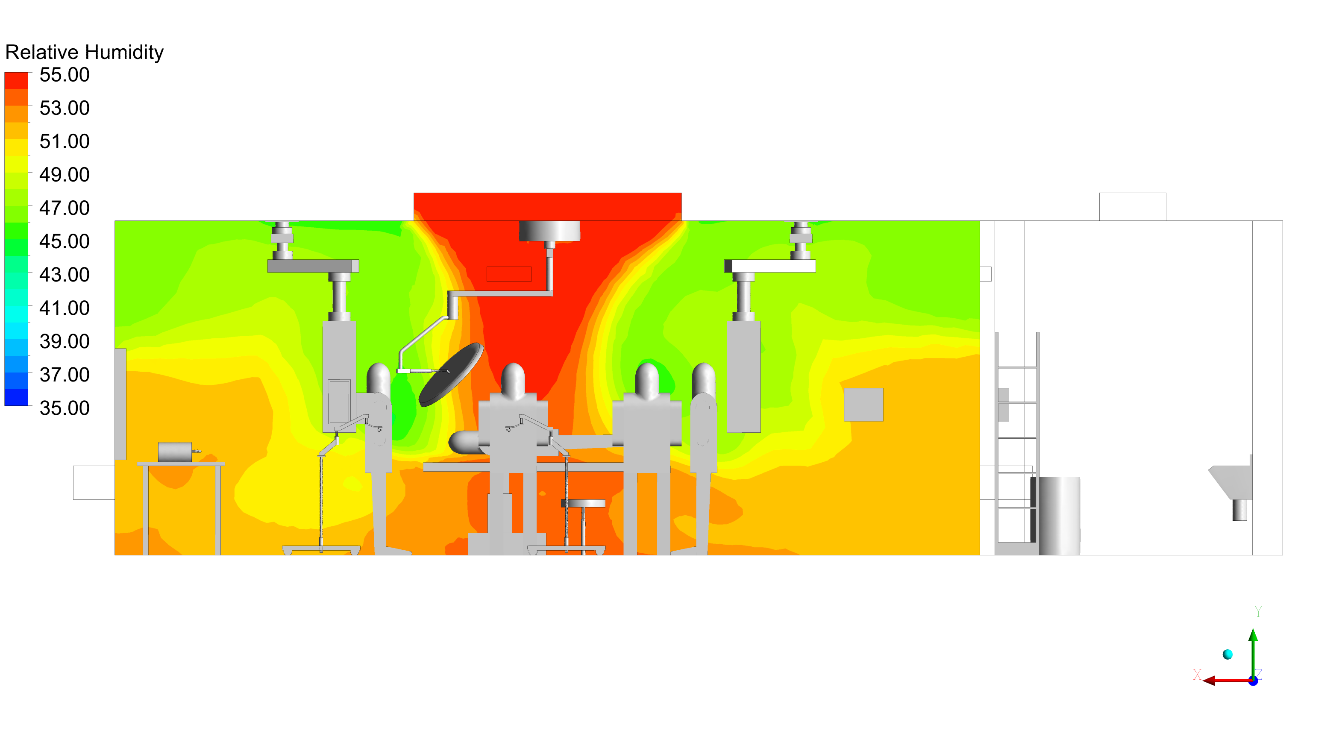
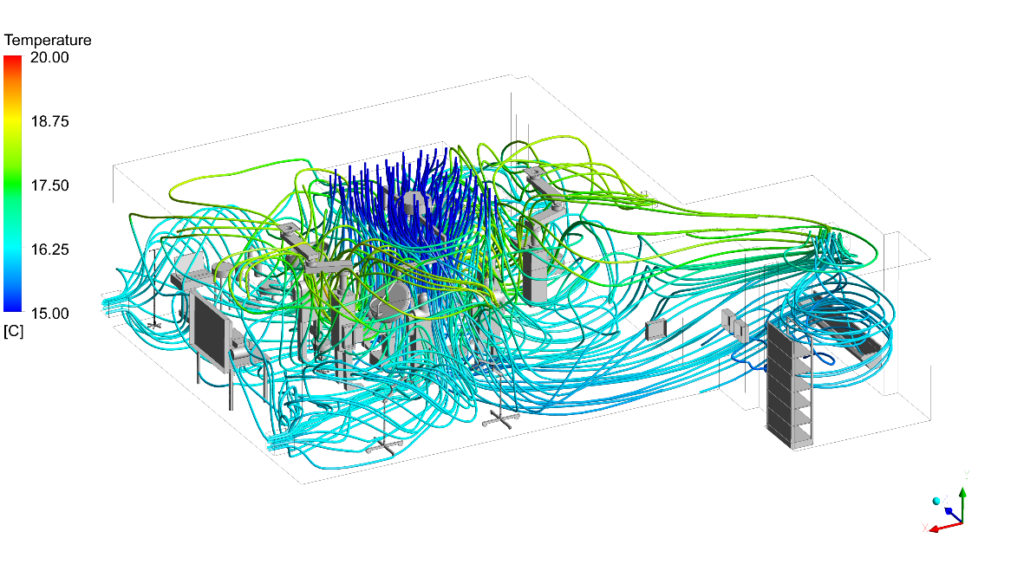
Why Conserve Solutions
Unlike conventional CFD studies that assess empty-room conditions, Conserve Solutions incorporates detailed geometry including equipment layout, staff positioning, and thermal loads. We evaluate recirculation zones, wake effects, and contamination pathways within the LAF region. This ensures airflow performance is validated where it matters most.
Client Outcome
Conserve’s CFD analysis gave our infection control team the evidence they needed to sign off the OT ventilation design with confidence. Knowing that ISO 5 cleanliness was confirmed at the surgical site – and that anaesthetic gas removal had been quantified, not assumed – removed the biggest clinical risk from our commissioning programme. The surgical light repositioning recommendation alone saved us from a particle count failure at handover.
Quick Links
Copyrights © 2026. Conserve Solutions. All Rights Reserved.
BIM Data, Cost & Handover Services
BIM Coordination & Constructability
BIM Modelling & Digital Delivery
Discipline BIM Modeling (Architectural, Structural, MEP)
BIM Automation & Digital Engineering
Structural & Steel Detailing Services
Architectural Design, Visualization & Presentation Support
BIM Modelling & Digital Delivery

